BEIJING, March 1 – Excessive fossil fuel consumption in recent decades has worsened environmental problems such as the greenhouse effect, driving urgency for technologies that capture and utilize carbon dioxide. One promising approach is the electrochemical CO2 reduction reaction (eCO2RR), which converts CO2 into valuable chemicals.
Alkali metal ions in the electrolyte are seen as critical to this process, influencing catalytic activity and product selectivity. But the mechanisms by which these cations regulate electrocatalytic reactions remain disputed, with researchers divided over the core determinants of their effect.
Most studies to date have focused on correlations between catalytic performance and qualitative spectral data, or on simplified models of electrode-electrolyte interfaces. While concentration and type of alkali metal cations have been widely examined, little work has addressed how their distribution patterns affect interfacial properties, kinetics and thermodynamics. The quantitative relationship between catalytic performance and cation behavior also remains unclear.
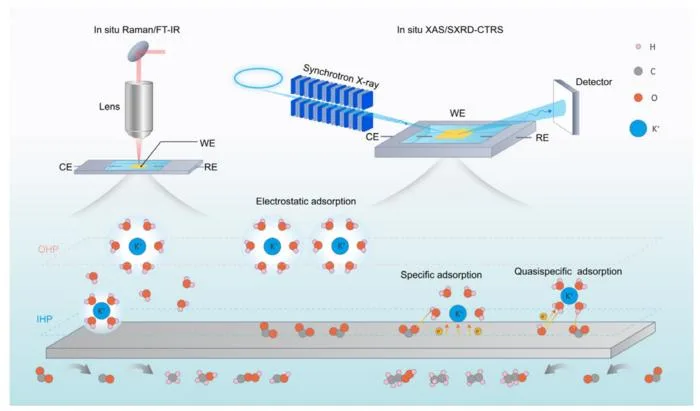
A team led by Prof. You-Nian Liu and Dr. Shanyong Chen of Central South University has reviewed recent advances in the field, publishing their findings in the Chinese Journal of Catalysis. The researchers outlined three distinct distribution patterns of alkali metal cations at the reaction interface – electrostatic adsorption, specific adsorption and quasi-specific adsorption – and examined how system variables influence these modes.
The study also explored the regulatory mechanisms of different adsorption modes on eCO2RR, clarified the physicochemical origins of the cation effect, and summarized the role of alkali metal cations across various electrolyte systems. In addition, the team assessed nitrogen-containing organic cations with alkali-like properties, highlighting their potential to assist or replace alkali metal cations in eCO2RR.