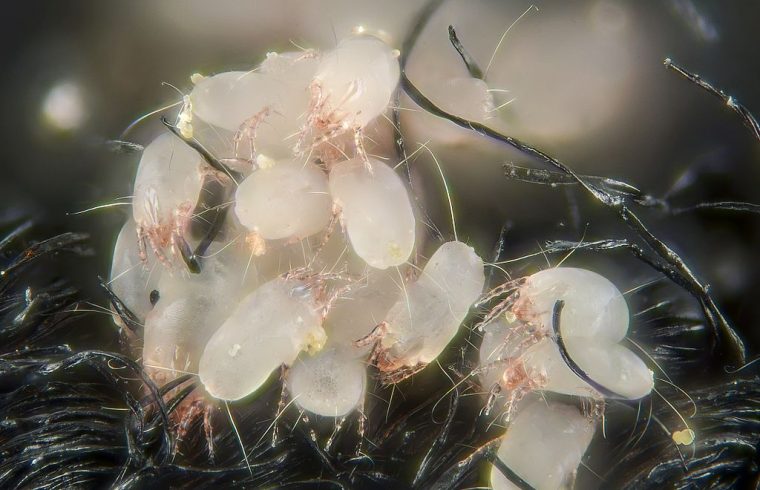
MADISON — Some of the world’s most common allergy-inducing critters, from dust mites to fungi to shellfish, have one thing in common: chitin, the polymer that makes tough cell walls in mushrooms, fungal spores and crunchy lobster exoskeletons.
A whiff of chitin triggers an immune response in the lungs, likely to prepare them to fend off fungal spores. But in some people, that reaction goes haywire, leading to dangerous inflammation and asthma. Despite these critical health consequences, scientists have not known just how the lungs sense and respond to chitin.
In a new study, University of Wisconsin–Madison researchers report discovering the first known receptor for chitin in mammals. Known as LYSMD3, the protein triggers an immune response in lung cells when it binds to chitin or chitin-bearing fungal spores.

The discovery of LYSMD3 solves one piece of the puzzle of the immune response to this common allergen, but more research is required to determine whether and how LYSMD3 may play a role in allergic reactions, including asthma.
“Chitin is ubiquitous in our environment. And it’s become apparent that chitin is a very potent allergic inflammatory stimulus,” says Bruce Klein, a UW–Madison professor of medical microbiology and immunology, who helped lead the new study. “But one of the unsettled questions has been what is the receptor that senses chitin? This has been something that the scientific community has been chasing for a long time.”
Klein, his former postdoctoral researcher and lead author Xin He, and their collaborators at other institutions published their findings July 20 in the journal Cell Reports.
Years ago, scientists discovered how plants sense chitin. So, He and his colleagues scanned the human genome for proteins that had chitin-binding portions similar to their plant cousins. They discovered four candidates, named LYSMD1 through LYSMD4. Only versions 3 and 4 were predicted to be located in the cell membrane and on its surface, where they might be able to interact with chitin.
To test whether the proteins could sense chitin, He experimentally reduced the amount of LYSMD3 and LYSMD4 in lab-grown human lung cells. Then he exposed the cells to chitin.
He found that the cells lacking LYSMD3 produced fewer inflammatory chemicals known as cytokines. Cytokines are the body’s first line of defense against invaders. LYSMD4 didn’t seem to respond to chitin.
“A key part of our study is that we used highly purified, soluble chitin oligomers,” says He. “We also applied a larger amount of chitin than the amounts usually used in experiments. When people have used low amounts of chitin, they might have overlooked receptors that have low affinity for chitin or low cell surface expression.”
In further experiments, the researchers completely deleted the LYSMD3 gene in lung cells. Like cells that produced less of the receptor, cells that produced no LYSMD3 at all secreted many fewer cytokines in response to chitin or chitin-bearing fungi.
The team also discovered that the purified LYSMD3 protein could tightly latch onto both purified chitin and natural chitin on fungal pathogens. And the protein was also able to bind to another component of fungal cell walls called beta-glucan, which might help it accurately detect invading pathogens.
Although LYSMD3 appears to play a role in sensing and responding to chitin in lung cells, it remains unclear whether it cooperates with other proteins or how it affects the intensity of the inflammatory response. That intensity is key to understanding when inflammation gets out of control to produce symptoms like asthma. Deciphering this process will require more research.
“Lots of this research was lab work with isolated human cells. We would like to know how the cells with this receptor behave in an intact animal, whether mouse or human,” says Klein. “One of the big questions for us is how does LYSMD3 influence its response in allergic inflammatory disorders such as asthma.”