SINGAPORE – New research from Duke-NUS Medical School, and the Agency for Science, Technology and Research identifies new anti-cancer treatment options.
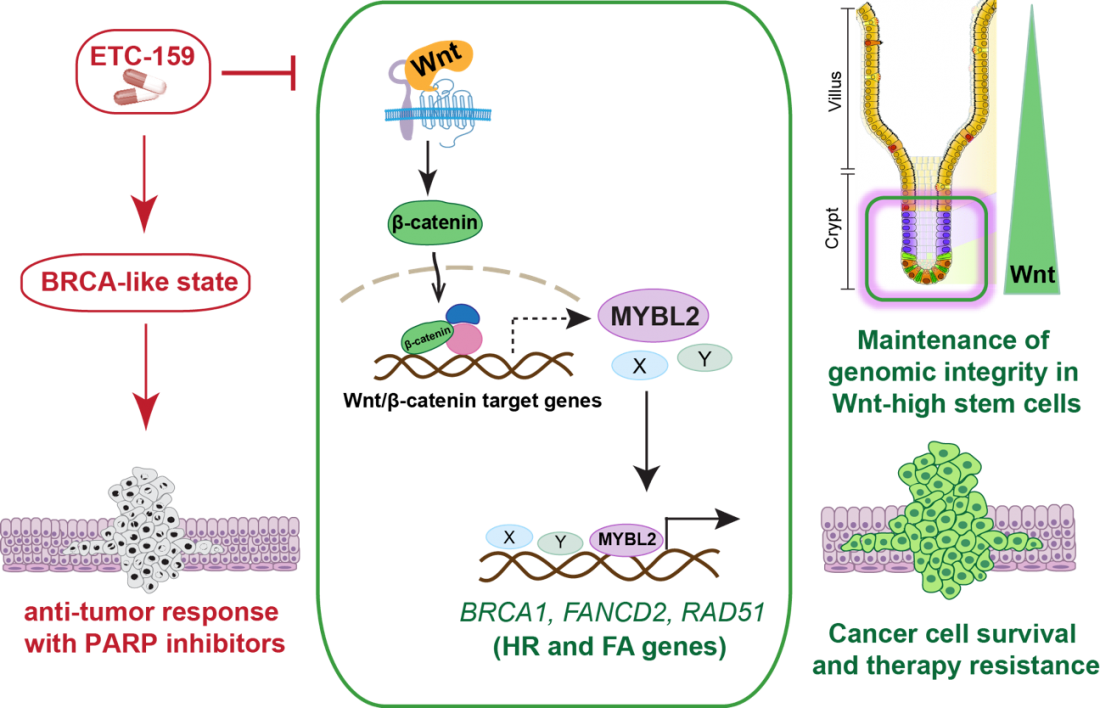
ETC-159, a made-in-Singapore anti-cancer drug that is currently in early phase clinical trials for use in a subset of colorectal and gynaecological cancers, could also prevent some tumours from resisting therapies by blocking a key DNA repair mechanism. Researchers from Duke-NUS Medical School and the Agency for Science, Technology and Research (A*STAR) in Singapore reported in the journal EMBO Molecular Medicine.
Among the many therapies used to treat cancers, inhibitors of the enzyme poly (ADP ribose) polymerase (PARP) prevent cancer cells from repairing naturally occurring DNA damage, including unwanted/harmful breaks in the DNA. When too many breaks accumulate, the cell dies.
“Some cancers have an overactive Wnt signalling pathway that may make them resistant to this sort of DNA damage,” said Assistant Professor Babita Madan, from Duke-NUS’ Cancer and Stem Cell Biology (CSCB) Programme and a senior author of the study. “Understanding how this pathway drives resistance to existing therapies could lead to the development of novel anti-cancer treatments.”
Normally, Wnt signalling proteins interact with cell receptors to activate the translocation of another protein, called beta-catenin, into the nucleus, where it regulates the activation of several genes.
“We found that, when Wnt signalling sends beta-catenin into the nucleus, it activates a family of DNA break repair genes,” said Professor David Virshup, director of the CSCB Programme and co-senior author of the study. “Cancers with excessive Wnt signalling, like colorectal cancer, therefore, have an enhanced ability to repair DNA breaks and thus escape the effects of PARP inhibitors.”
The team found that blocking Wnt signalling with a drug called ETC-159 reversed PARP inhibitor resistance in several cancer cell lines.
ETC-159 inhibits an enzyme called porcupine, which in turn, prevent the secretion of Wnt proteins. ETC-159 is being tested in a clinical trial for use in cancers with overactive Wnt signalling, amongst other therapeutic indications
Analysis of this pre-clinical study shows that therapeutic doses of ETC-159 appear to be well tolerated by the gut, without causing toxicity. This means that a low dose of ETC-159, when given alongside PARP inhibitors, could prevent cancer resistance to treatment with PARP inhibitors while sparing intestinal stem cells, providing further options for treating cancers with hyperactive Wnt signalling.
Through this study, the researchers also learned that the same signal for DNA repair helps to prevent mutations from developing in stem cells residing inside the intestinal epithelium, further confirming the importance of normal Wnt signalling in stem cell maintenance.
ETC-159 was jointly developed by Duke-NUS and the Experimental Drug Development Centre (EDDC), a national platform for drug discovery and development hosted by A*STAR. The Wnt-pathway inhibitor is a novel small-molecule drug candidate that targets a range of cancers. It is currently progressing through clinical trials as a treatment for a subset of colorectal and gynaecological cancers.
“These findings improve our understanding of how Wnt signalling enhances DNA repair in stem cells and cancers, maintaining their genomic integrity,” said Dr May Ann Lee, a group head at EDDC and also a senior author of the study. “Conversely, interventions that block Wnt signalling could cause some cancers to be more sensitive to radiation and other DNA damaging agents.”