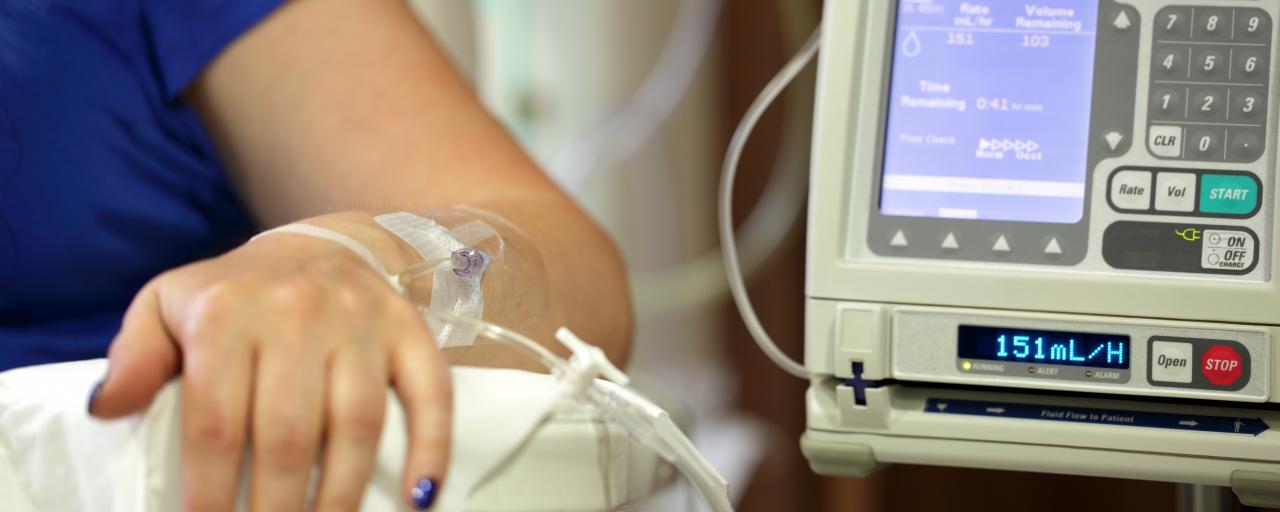
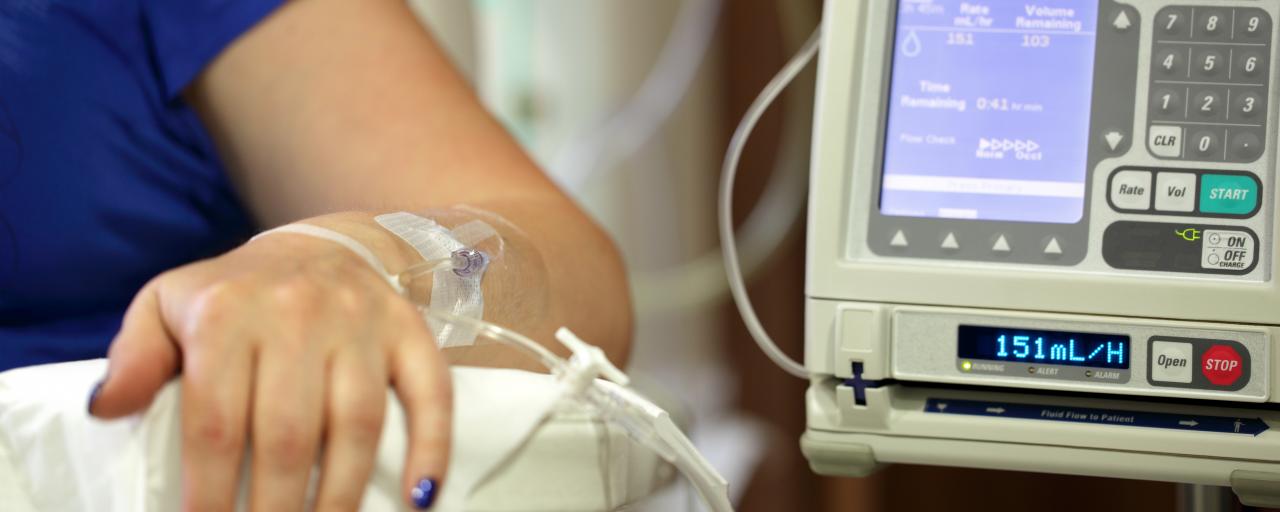
 Heron Therapeutics Inc.’s injectable therapy, Sustol (granisetron), for chemotherapy-induced nausea and vomiting (CINV) was approved by the U.S. Food and Drug Administration Wednesday, August 11.
Heron Therapeutics Inc.’s injectable therapy, Sustol (granisetron), for chemotherapy-induced nausea and vomiting (CINV) was approved by the U.S. Food and Drug Administration Wednesday, August 11.
Sustol is the first approved product that uses Heron’s Biochronomer drug-delivery technology, which maintained effective levels of the medication for at least five days, covering the acute and delayed phases of CINV. A global, Phase 3 program, composed of two, large clinical trials, evaluated Sustol’s efficacy and safety in more than 2,000 patients with cancer.
[pullquote]“Despite advances in the management of CINV, up to half of patients receiving chemotherapy can still experience CINV, with delayed CINV being particularly challenging to control,” said Dr. Ralph Boccia.[/pullquote]
The newly approved drug is a serotonin-3 (5-HT3) receptor antagonist used in combination with other antiemetics.
Sustol had been rejected twice previously by the FDA. In 2010, Heron (then called A.P. Pharma) received its first rejection for Sustol. The company returned with additional data, but three years later, the drug was again rejected due to manufacturing and data analysis problems.
“Despite advances in the management of CINV, up to half of patients receiving chemotherapy can still experience CINV, with delayed CINV being particularly challenging to control,” said Dr. Ralph Boccia, medical director at the Center for Cancer and Blood Disorders, in the release. “In our experience, other 5-HT3 receptor antagonists, including palonosetron, are generally effective for 48 hours or less.”
CINV, one of the most acute side effects of chemotherapy, occurs in up to 80 percent of patients and has a significant impact on a patient’s quality of life, according to the National Cancer Institute.