(RS)— High-entropy alloys (HEA) have a unique chemical composition that makes them strong, ductile, and resistant to wear and tear even at high temperatures. Yet this chemical composition also makes them difficult to study. Now, a collaborative research team has created a new experimental platform that enables the control of the atomic-level structure of HEAs’ surfaces and the ability to test their catalytic properties.
Introduced in 2004, high-entropy alloys (HEAs) are alloys composed of multiple principal elements in nearly equiatomic proportions. Their unique chemical composition results in a high degree of chemical disorder, i.e. entropy, and produces remarkable properties such as high strength, ductility, and strong wear-and-tear resistance even at high temperatures. Scientists have dedicated a significant amount of attention to developing novel HEAs to help improve the performance of various electrocatalyst materials.
Because they are made up of differing constituent elements, HEAs’ atomic-level surface designs can be complex. But unraveling this complexity is crucial since the surface properties of materials often dictate their catalytic activity. Hence why researchers are seeking to understand the correlation between the atomic arrangement and the catalytic properties exhibited by HEAs.
Now, a collaborative research team has created a new experimental platform that enables the control of the atomic-level structure of HEAs’ surfaces and the ability to test their catalytic properties. Their breakthrough was reported in the journal Nature Communications on July 26, 2023.
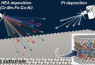
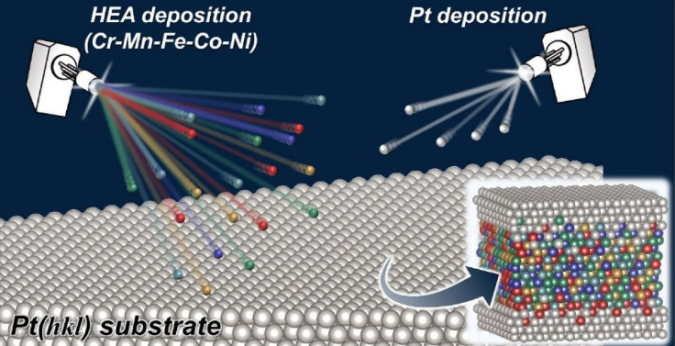
“In our study we made thin layers of an alloy called a Cantor alloy, which contains a mix of elements (Cr-Mn-Fe-Co-Ni), on platinum (Pt) substrates,” explains Toshimasa Wadayama, co-author of the paper and a professor at Tohoku University’s Graduate School of Environmental Studies. “This produced a model surface for studying a specific reaction called the oxygen reduction reaction (ORR).”
Using advanced imaging techniques, the group examined the atomic-level structure of the Pt-HEAs’ surfaces and studied their ORR properties. They discovered that the Pt-HEAs’ surfaces performed better in ORR compared to surfaces made of a platinum-cobalt alloy. This indicates that the atomic arrangement and distribution of elements near the surface, which creates a ‘pseudo-core-shell-like structure,’ contributes to the excellent catalytic properties of Pt-HEAs.
Wadayama and his group stress the wide applicability of their findings, both for any constituent elements and to other nanomaterials.
“Our newly constructed experimental study platform provides us with a powerful tool to elucidate the detailed relationship between multi-component alloy surface microstructures and their catalytic properties. It is valid for clarifying the precise correlations among the atomic-level, surface microstructure, and electrocatalytic properties of HEAs of any constituent elements and ratios and, thus, would provide reliable training datasets for materials informatics. The platform is applicable not only to electrocatalysis but also in various fields of functional nanomaterials.”
Looking ahead, the group hopes to expand this platform into practical electrocatalysis by using Pt-HEA-nanoparticles that seek to increase electrochemical surface areas.