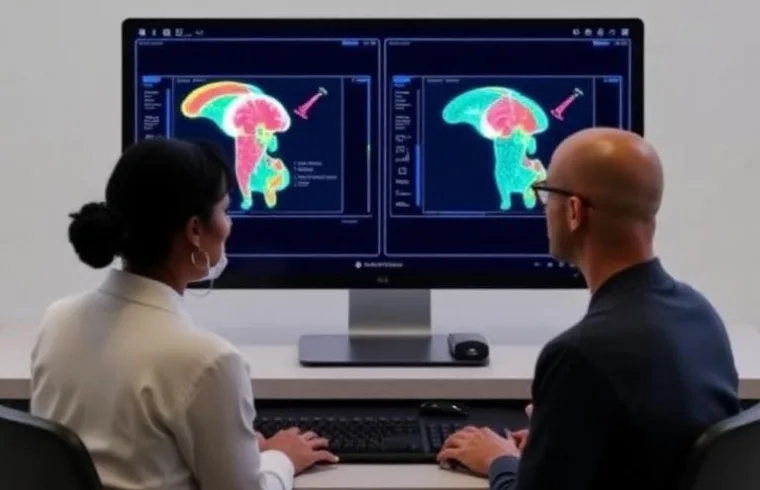
BARCELONA, March 23 — Clinical trials, the backbone of modern medicine, may be entering a new digital era.
A review published in Reviews on Recent Clinical Trials by Francisco Epelde, Dolores Rexachs, Álvaro Wong, and Emilio Luque examines the growing role of Virtual Clinical Trials (VCTs). These computer-based simulations test drugs and therapies without enrolling human patients.
Digital simulations
The study, conducted by researchers at Parc Taulí Hospital Universitari and the Universitat Autònoma de Barcelona, highlights how VCTs use mathematical models, artificial intelligence, and large datasets to create “virtual populations.”
These digital patients can represent different ages, genetic traits, and disease profiles, allowing researchers to explore scenarios that would be costly or ethically difficult in traditional trials.
Speed and cost
Traditional trials often cost hundreds of millions of dollars and take years to complete. Virtual trials, by contrast, can deliver results in days.
The authors say this speed could prove critical during pandemics or fast-moving health crises, where rapid decisions save lives. Simulations also allow researchers to identify weak drug candidates earlier, reducing risks to human participants.
Ethical concerns
The paper warns that VCTs are not a replacement for human studies. Models depend on the quality of underlying data and may miss rare side effects or oversimplify patient responses.
“Virtual trials must be validated against real-world evidence,” the authors note, stressing that patient experiences and quality-of-life outcomes cannot be replicated by algorithms alone.
Equity and bias
Advocates say VCTs could broaden representation by including groups often underrepresented in clinical research.
However, the review cautions that biased or incomplete datasets risk reproducing inequalities inside simulations. Ensuring diversity requires transparent assumptions and ongoing auditing.
Regulation
Existing regulatory frameworks were designed for human-based trials and do not easily apply to digital simulations.
The authors call for updated standards that prioritize transparency, validation, and safeguards to trigger human testing when uncertainty is high. Ethical review boards, they argue, must adapt to evaluate algorithms as well as protocols.
Hybrid future
The review concludes that the future of clinical research will be hybrid, combining virtual trials, digital twins, synthetic control arms, and traditional studies. Used responsibly, VCTs could accelerate drug development and reduce risks. Used carelessly, they could amplify bias and erode trust.