Magnesium rechargeable batteries (MRBs), where high-capacity Mg metal is used as the anode material, are promising candidates for next-generation batteries due to their energy density, safety, and cost. However, the lack of high-performance cathode materials impedes their development.
Like their lithium-ion counterparts, transition metal oxides are the staple cathode materials in MRBs. Yet the slow diffusion of Mg ions inside the oxides poses a serious problem. To overcome this, some researchers have explored sulfur-based materials. But sulfur-based cathodes for MRBs have severe limitations: low electronic conductivity, sluggish Mg diffusion in solid Mg-S compounds, and dissolubility of polysulfides into electrolytes, which results in low-rate capability and poor cyclability.
Now, a research team that included Tohoku University’s Dr. Shimokawa and Professor Ichitsubo has developed liquid-sulfur/sulfide composite cathodes enabling high-rate magnesium batteries. Their paper has been published in the Journal of Materials Chemistry A.
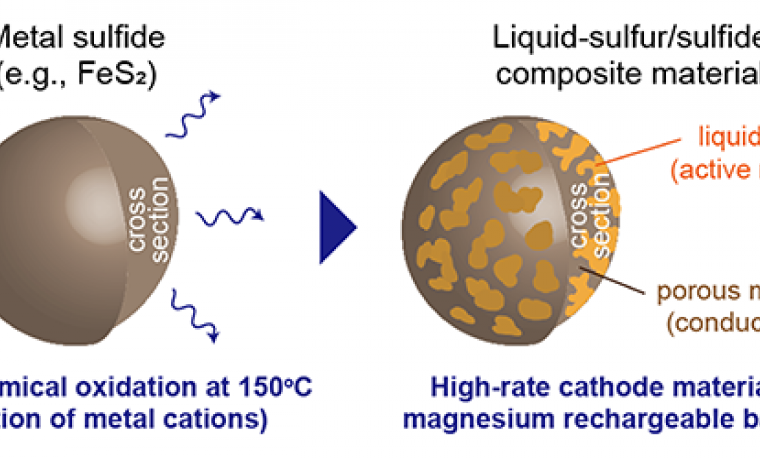
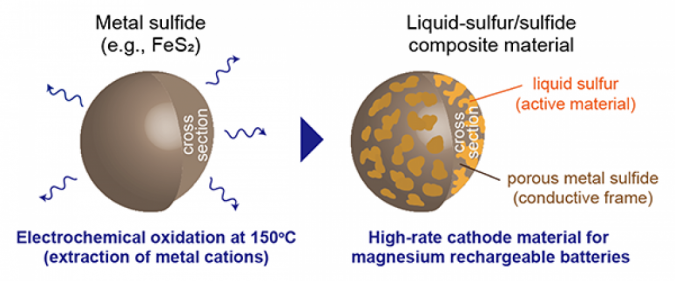
The liquid-sulfur/sulfide composite materials can be spontaneously fabricated by electrochemically oxidizing metal sulfides, such as iron sulfide, in an ionic liquid electrolyte at 150. The composite material showed high performance in capacity, potential, cyclability, and rate capability.
The researchers achieved the discharge capacity of ~900 mAh/g at a high current density of 1246 mA/g based on the mass of active sulfur. In addition, they revealed that the discharge potential was enhanced by utilizing non-equilibrium sulfur formed by fast charging processes.
This material allowed for a stable cathode performance at 150 for more than 50 cycles. Such a high cyclability could be attributed to the following points: high structural reversibility of the liquid state active material, low solubility of polysulfides into the ionic liquid electrolyte, and high utilization ratio of sulfur due to its adhesion to conductive sulfide particles that form a porous morphology during the synthesis of the composite materials.
Despite the researchers’ progress, several problems remain. “We need electrolytes that are compatible with both the cathode and anode materials because the ionic liquid used in this work passivates the Mg-metal anode,” said Shimokawa. “In the future, it is important to develop new electrochemically stable electrolytes to make MRBs more practical for widespread use.”
Although MRBs are still in the development stage, the research team is hopeful their work provides a new way to utilize liquid sulfur as high-rate cathode materials for MRBs. “This would boost the improvement of sulfur-based materials for achieving high-performance next-generation batteries,” added Shimokawa.